Oxidation state, on the other hand, is a way of keeping track of the number of bonds between atoms in a molecule and is calculated by adding the number of covalent bonds between the atoms and subtracting the number of lone electrons.ĪLSO READ: Difference Between Nitromethane And Methyl Nitrite Formal charge is a way of keeping track of the electrons in a molecule, and is calculated by subtracting the number of valence electrons from the number of electrons assigned to each atom in the molecule. Keeping track of formal charge and oxidation state can be a challenge for chemists, as they are two different concepts that are related but distinct. Tips for keeping track of formal charge and oxidation state Therefore, oxidation state is a more accurate measure of an atom’s charge than formal charge. Oxidation state, on the other hand, looks at the total number of electrons an atom has, and assigns a charge based on the total number of electrons an atom has gained or lost. The main difference between formal charge and oxidation state is that formal charge is a bookkeeping tool used to keep track of electrons, while oxidation state is a measure of the oxidation number of an atom in a molecule.įormal charge takes into account the number of valence electrons on an atom, and assigns a charge based on how many electrons an atom has gained or lost. While both formal charge and oxidation state are related to the charge of an atom in a molecule, they are not the same. Common misconceptions about formal charge and oxidation stateįormal charge and oxidation state are both important concepts when it comes to understanding chemistry, but they are often confused for one another. In essence, formal charge is the net difference between the number of electrons that an atom has, while oxidation state is the net number of electrons lost or gained. On the other hand, oxidation state is the number of electrons a atom has lost or gained in a reaction. Formal charge is the difference between the number of valence electrons of an atom in a molecule and the number of electrons the atom is actually holding.ĪLSO READ: Difference Between Acetylene Black And Carbon Black 
While both are related to electron transfer, there are some key differences to keep in mind. Examples of formal charge and oxidation stateįormal charge and oxidation state can be two of the most confusing aspects of chemistry to understand. When calculating formal charge and oxidation state, it’s important to keep these factors in mind. The oxidation state of an atom can also be affected by the overall polarity of a molecule. There are several factors that affect the formal charge and oxidation state of an atom, including the number of electrons in the atom’s valence shell, the number of lone pairs in the atom’s outer shell, and the number of bonds the atom has with atoms of different elements. The oxidation state, on the other hand, is the charge an atom would have if all the bonds to atoms of different elements were to be 100% ionic. 
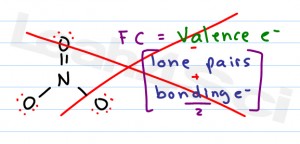
The formal charge of an atom is the charge on an atom in a molecule or ion, calculated by subtracting the number of electrons surrounding the atom from the number of protons in the nucleus. This suggests that option A is the correct answer.When it comes to understanding formal charge and oxidation state, it’s important to know the difference between the two. The formal charges on the nitrogen atoms in the case of azide ion from left to right are -1, +1, -1. Hence the central nitrogen atom of the azide ion has a +1 formal charge. The formal charge on the central nitrogen atom = The bonding electrons and nonbonding electrons in the central nitrogen atom of azide are eight and zero, respectively. The valence electrons in the N atom are five. This suggests that nitrogen atoms at the terminal positions of azide ion have a -1 formal charge.Ĭalculation of formal charge for the central nitrogen atom of azide ion. Putting these values in the above equation.įormal charge on terminal nitrogen atoms = -1. The bonding and nonbonding electrons in the terminal nitrogen atom of the azide ion are four. Hence N has five electrons in the valence shell. The electronic configuration of the N atom is 1s² 2s² 2p³. This suggests that the formal charge on these nitrogen atoms will also same. The terminal nitrogen atoms of the azide ion have the same chemical environment. Formal charge = (number of valence electrons) - (number of nonbonding electrons) - (number of bonding electrons) 2 \textįirst, calculate the formal charge on terminal Nitrogen atoms of azide ion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
